Description
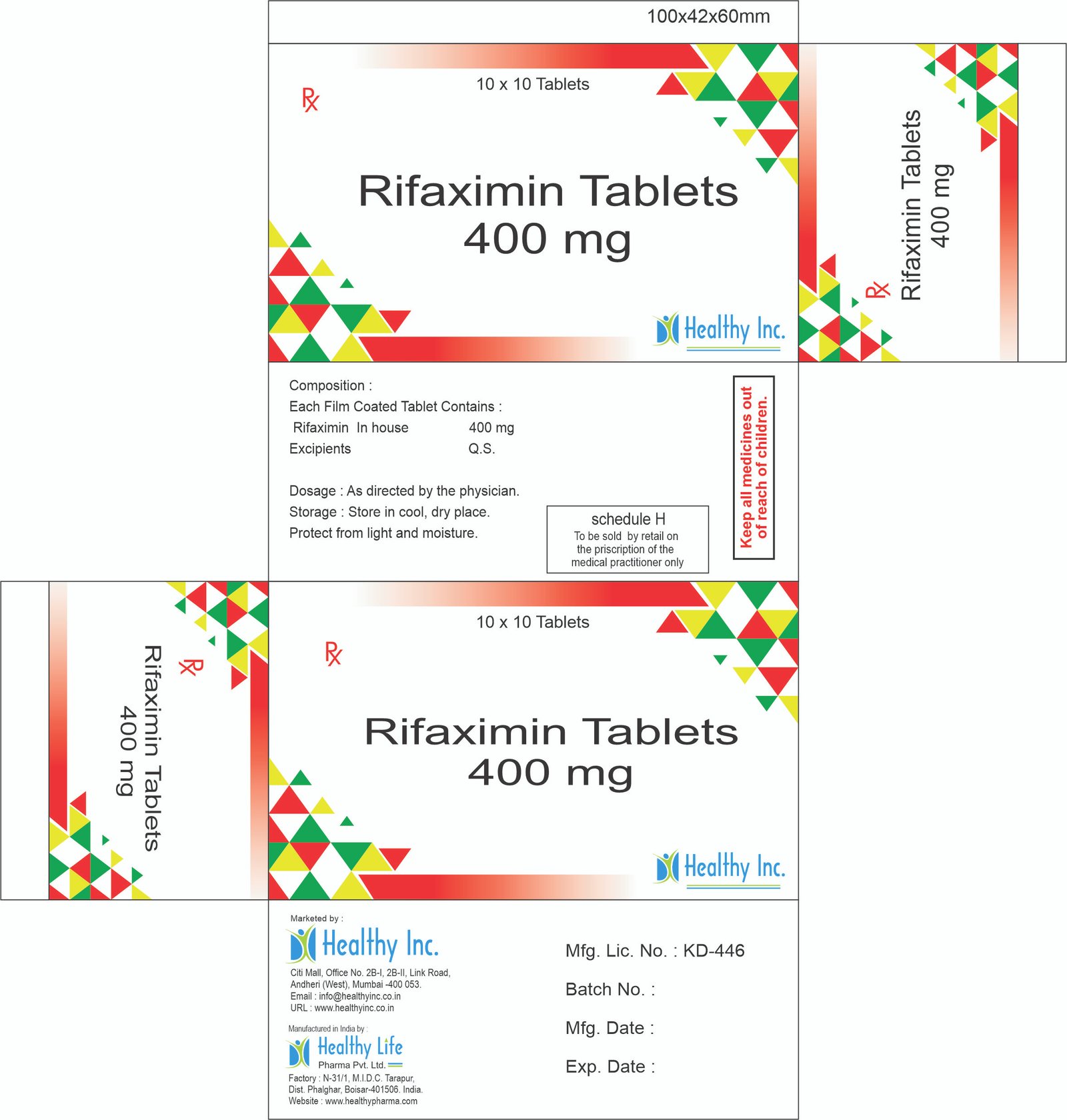
Rifaximin Tablets (200mg / 400mg / 550mg)
Manufactured by: Healthy Life Pharma Pvt. Ltd. (WHO-GMP Certified)
Exported by: Healthy Inc (Star Export House)
1. Product Introduction
Healthy Life Pharma Pvt. Ltd. is a specialized Manufacturer of Rifaximin Tablets in India. Rifaximin is a unique, non-systemic Rifamycin Antibiotic. Unlike other antibiotics that are absorbed into the bloodstream, Rifaximin acts locally in the gastrointestinal tract, making it the “Gold Standard” for treating Traveler’s Diarrhea, Hepatic Encephalopathy, and Irritable Bowel Syndrome with Diarrhea (IBS-D).
We offer Contract Manufacturing (Third Party) services for this high-value gastroenterology product. Rifaximin presents significant manufacturing challenges due to its polymorphic nature (crystal forms affect absorption). Our WHO-GMP certified facility in Mumbai utilizes specialized polymorph-screening technologies to ensuring we deliver the biologically active form. Healthy Inc manages the export logistics, supplying major hospital chains and gastroenterology centers across the USA, Europe, the Middle East, and Latin America.
2. Product Specifications
| Parameter | Specification |
| Product Name | Rifaximin Tablets |
| Generic Name | Rifaximin Tablets USP / BP / IP |
| CAS Number | 80621-81-4 |
| Strength | 200mg / 400mg / 550mg |
| Dosage Form | Film Coated Tablet (Pink/Red, Capsule-shaped) |
| Standard | USP / BP / IP Compliant |
| Therapeutic Class | Gastrointestinal Antibiotic |
| Shelf Life | 36 Months |
| Packaging | 10×10 Blister / 1×10 Blister / Alu-Alu Pack |
3. Manufacturing Technology
Polymorph control is the critical quality attribute.
The Manufacturer: Healthy Life Pharma Pvt. Ltd.
Polymorph Specificity: Rifaximin exists in multiple crystal forms ($\alpha, \beta, \gamma, \delta, \epsilon$). The $\alpha$ (Alpha) form is non-absorbable (systemic absorption < 0.4%), which is desired for local gut action. Other forms can be absorbed systemically, which increases toxicity risks. We strictly control the crystallization and drying process to ensure only the safe, non-absorbable form is produced.
Solubility Profile: Although it is not meant to be absorbed, it must dissolve in the gut fluid to kill bacteria. We use Micronization to increase the surface area, ensuring it coats the intestinal lining effectively.
Direct Compression: We utilize direct compression techniques where possible to minimize heat and moisture exposure, which can induce polymorphic shifts.
The Exporter: Healthy Inc
High-Barrier Packaging: We recommend Alu-Alu Blisters to prevent moisture ingress, as moisture can cause the crystal structure to shift from the desired form to a less stable one during storage.
4. Quality Assurance
We adhere to strict Pharmacopoeial standards:
X-Ray Diffraction (XRD): We perform XRD analysis on the API and finished formulation to validate that the crystal structure (Polymorph Alpha) has not changed during manufacturing.
Dissolution: We test release profiles to ensure consistent local availability in the intestine.
Assay: We confirm the potency is strictly within 90-110% of the label claim.
Related Substances: We monitor for Rifamycin-related impurities to ensure safety.
5. Why Use Rifaximin?
It kills gut bacteria without side effects elsewhere in the body.
Mechanism: It binds to the beta-subunit of bacterial DNA-dependent RNA polymerase, blocking transcription. It is active against E. coli (enterotoxigenic strains), Salmonella, Shigella, and ammonia-producing gut bacteria.
Key Indications:
Traveler’s Diarrhea: Caused by non-invasive strains of E. coli.
Hepatic Encephalopathy: Reduces blood ammonia levels by killing ammonia-producing bacteria in the gut, preventing confusion/coma in liver failure patients. (Dosage: 550mg BID).
IBS-D: Relieves bloating and diarrhea in Irritable Bowel Syndrome by treating Small Intestinal Bacterial Overgrowth (SIBO).
6. Export and Regulatory Support
We streamline the registration process for our B2B partners:
Dossier Support: We offer CTD and ACTD Dossiers (with XRD Polymorph Data) for quick registration.
Certificates: Free Sale Certificate (FSC), COPP (WHO-GMP), and COA.
Logistics: Efficient shipping via Air or Sea (FOB Mumbai / CIF).
7. Frequently Asked Questions
Q: Who manufactures Rifaximin Tablets?
A: Healthy Life Pharma Pvt. Ltd. manufactures them in India.
Q: Is it safe for kidneys?
A: Yes. Because < 0.4% of the drug enters the bloodstream, it has virtually no systemic toxicity and does not burden the kidneys or liver like other antibiotics.
Q: Can it treat a UTI?
A: NO. Rifaximin stays in the gut. It does not reach the urinary tract, lungs, or skin. It is only for intestinal infections.
Q: Does it cause antibiotic resistance?
A: Rifaximin does not carry plasmids for resistance, meaning the risk of developing resistant bacteria is very low compared to Ciprofloxacin or Azithromycin.
CLINICAL PHARMACOLOGY & SAFETY INFORMATION
(For Registered Medical Practitioners & Patient Reference)
8. Dosage and Administration
Traveler’s Diarrhea: 200mg three times daily for 3 days.
Hepatic Encephalopathy: 550mg twice daily (long-term).
IBS-D: 550mg three times daily for 14 days.
Administration: Swallow whole. Can be taken with or without food.
9. Side Effects and Precautions
Common Side Effects: Nausea, flatulence, headache, and abdominal pain (often hard to distinguish from the symptoms of the disease itself).
Superinfection: Prolonged use may result in fungal or C. difficile overgrowth (though rare).
Interactions: Minimal drug interactions due to lack of systemic absorption. P-glycoprotein inhibitors (like Cyclosporine) may slightly increase Rifaximin absorption.
10. Storage Instructions
Store below 25°C in a dry place.
Protect from moisture. (Crucial for polymorph stability).